Feringa’s motors in the news
Chemical & Engineering News (a publication of the American Chemical Society) recently published two articles on papers by Ben Feringa: one about a muscle that is flexed by molecular motors and the other about a new environmentally friendly process that makes useful building blocks for plastic and pills.
They say that winning the Nobel Prize is the kiss of death for your research, but a year after receiving this highest scientific accolade, Ben Feringa is still blazing a trail. We already reported on the article in which he describes how an antibioticum can be switched on and off with visible light. This appeared in Science Advances and was discussed in an article in Nature News.
Enthusiasm

Now two publications from Feringa’s lab have been featured on the website of Chemical & Engineering News. The first (5 December) is about an artificial muscle that responds to light by flexing. The muscle consists of motor molecules that self-assemble into fibres, forming a hydrogel. Researchers from Feringa’s group thus created a water-soluble version of the standard light-activated motor. In response to ultraviolet light the gel, which is composed of 95% water, can lift a 400 mg piece of paper. A molecular motion is thus amplified into a macroscopic motion.
Specialists in the field of artificial muscles are enthusiastic about this aqueous motor muscle. These are generally composed of polymers, but Feringa’s discovery could easily play a role in biological systems or soft robotics.
Sustainable
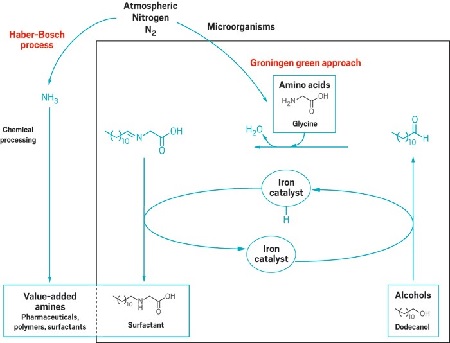
The second article (featured on C&E News on 11 December) is about Feringa’s other field of expertise: catalysis . It pertains to making alkylated amines . These are important building blocks for the chemical industry, which uses them to make plastics or medicine, for instance. The drawback is that the alkylation requires rather a lot of energy (it occurs at 500 degrees Celsius), does not use renewable materials and produces waste.
The new method developed by Feringa, his colleague Katalin Barta and her PhD student Tao Yan uses amino acids (a renewable material) harvested from bacteria and relies on a simple reaction to alcohol. The only ‘waste’ product is water. The catalyst initially used for this contained the metal ruthenium, but this could be replaced by everyday iron.
Director of the Center for Green Chemistry & Green Engineering at Yale University in the United States, Paul T. Anastas, calls the discovery ‘nothing but revolutionary’. It would make it cheaper and cleaner to produce important building blocks for the chemical industry. The Groningen researchers have applied for a patent for the new catalytic process.
| Last modified: | 17 March 2020 2.29 p.m. |
More news
-
10 June 2024
Swarming around a skyscraper
Every two weeks, UG Makers puts the spotlight on a researcher who has created something tangible, ranging from homemade measuring equipment for academic research to small or larger products that can change our daily lives. That is how UG...
-
21 May 2024
Results of 2024 University elections
The votes have been counted and the results of the University elections are in!

